What is photosynthesis?
Photosynthesis literally means synthesis (or manufacturing) with light.
It is the physiological process occurring in plants by which carbon dioxide (CO2) and water (H2O), in the presence of visible light and chlorophyll, are converted to organic compounds which store chemical energy.
It also occurs in other chlorophyll-containing organisms such as algae and cyanobacteria.
In 1993, Howard Gest of Indiana University gave the following definition of photosynthesis: Photosynthesis is a series of processes in which electromagnetic energy is converted to chemical energy used for biosynthesis of organic cell materials; a photosynthetic organism is one in which a major fraction of the energy required for cellular synthesis is supplied by light(Gest 2002).
There are many reasons why it is important to understand what is photosynthesis.
In fact, this Physico-chemical process (Govindjee 2000) is a necessary subject in plant physiology, specifically in relation to crop agriculture.
To enhance crop productivity, the process of photosynthesis must be well understood.
It is so because this process is the main reason why plants continue to survive, grow, and produce economic organs such as the roots, stems, leaves, flowers, fruits, and seeds that humans use in various ways.
Plants are likewise the ultimate source of energy and nutrition for animals and other forms of life which are incapable of directly harvesting and utilizing the energy from the sun.
In fact, almost all forms of life depend on photosynthesis for their supply of energy.
The word “almost” is used because some bacteria obtain energy from H2S or H2 in complete darkness (Mathews and Van Holde 1990).
Without photosynthesis, there will be no plants to form the food pyramid.
And so because man’s survival hinges on photosynthesis, every human ought to be aware of at the least be able to give one simple answer to the question What is Photosynthesis?
Note: Do you know that the one who himself coined photosynthesis insisted on a different word? Click here to read.
More on What is Photosynthesis
Functions in Photoautotrophs
Photosynthesis is essential to plants and other photoautotrophs, those organisms which have the rare ability to utilize sunlight as a source of energy.
It is through this process that they harvest solar energy and convert it into chemical energy.
This chemical energy in turn is used in order to perform work such as biosynthesis, translocation, and reproduction.
Without photosynthesis, there will be no way to utilize the energy from the sun.
Without photosynthesis, no plant will exist and this will have dire consequences for life on Earth.
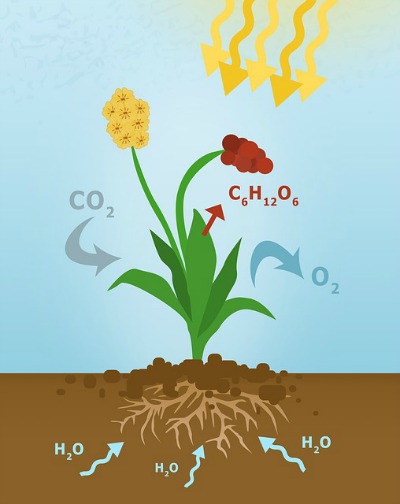
With rare exceptions as in albino plants, all plants produce their own food through photosynthesis from which chemical energy is extracted when needed.
The carbon that is fixed from the atmosphere in the process is also utilized as structural bases in the synthesis of organic compounds necessary for growth and development.
Importance to Heterotrophs
Photosynthesis is likewise essential to heterotrophs as a source of energy.
Heterotrophic organisms, such as humans and animals, are those non-photosynthesizing organisms that are incapable of utilizing directly the energy from the sun.
Without the natural apparatus to capture solar energy and convert it to chemical energy for their own use, heterotrophs must necessarily obtain it from those trapped in the organic compounds in photosynthetic organisms or other organisms that have harvested this energy from plants and plant-eaters.
They derive this energy from the food or feed that they consume in the same way that vehicles are powered by the energy stored in fuels and made available in usable form through the process of combustion.
These organic compounds that serve as a depository of chemical energy consist of carbohydrates, proteins, nucleic acids, and lipids.
In relation to human nutrition, Wardlaw et al. (2004) provide the following average energy content of these compounds: carbohydrates- 4 kcal/g, protein- 4 kcal/g, fats, and oils- 9 kcal/g.
Supply of Oxygen for Aerobic Organisms
In addition, photosynthesis is also vital to oxygen-requiring or aerobic organisms because the process generates oxygen in the photolysis of water during the light reactions.
Oxygen is released into the atmosphere and is utilized by aerobic organisms including plants themselves in the process of respiration.
However, the most dominant supply of oxygen on Earth comes from algae.
Such a type of photosynthesis in which oxygen is generated is called oxygenic photosynthesis.
Fossil Fuels for Human Use
Another aspect in relation to what is photosynthesis is in connection with man’s daily chores and his enjoyment.
Although it is not apparent, photosynthesis has made modern life easier.
Let there be light and indeed, artificial light using electrical energy has become readily available; industrial factories have made available processed food and other products for human use; we ride in cars and go to distant places in trains, ships and planes; electrical gadgets and devices give us comfort at home regardless of the season and time of the day.
Photosynthesis made these possible by producing the energy stored in petrochemicals and other fossil fuels.
Coal, oil, and natural gases are prehistoric plants and animals that have undergone decomposition over millions of years (US-DOE 2012).
In addition, the oxygen that is evolved in the process of photosynthesis is used in the combustion or burning of fuel.
Peat, partially decomposed moss plants like those which have accumulated for thousands of years in peat bogs in northern Europe, have an energy value equivalent to 3,300 calories per gram.
This value exceeds that of wood but only half of the coal.
The volume of peat in the United States exceeds 60 billion tons which is equivalent to about 240 billion barrels of oil.
In countries under the former Soviet Union, about 80 power plants that generate electricity have been maintained using as fuel more than 200 million tons of peat harvested annually (Moore et al. 2003).
What is Photosynthesis: Advances
Researches are still continuing and researchers are close to developing the technology on human-made photosynthesis. Steinberg-Yfrach et al. (1998, cited by Govindjee 2000) succeeded in producing “lots of ATP” artificially with synthetic energy.
Presently, there are ongoing researches that can revolutionize food and energy production.
One consists of creating an “artificial leaf” that is capable of collecting solar energy and converting it to liquid fuel by applying similar photosynthetic reactions (Biotechnology and Biological Sciences Research Council 2012).
Certainly, the outcomes of these investigations will broaden further the picture of what is photosynthesis.
REFERENCES
Biotechnology and Biological Sciences Research Council. 2012. Human-made photosynthesis revolutionized food and energy production. Retrieved Feb. 26, 2012, from http://www.sciencedaily.com/releases/2012/02/120217145755.htm.
GEST H. 2002. Definition of photosynthesis: History of the word photosynthesis and evolution of its definition. Photosynthesis Research. 73: 7–10. Retrieved June 17, 2012, from http://nsm1.nsm.iup.edu/nuttle/Botany%20Spring%202011/3.%20Gest%20%282002%29%20 Photosynthesis%20Research%2073%207%20-%20History%20of%20phs%20research.pdf.
GOVINDJEE. 2000. Milestones in photosynthesis research. In: Yunus M, Pathre U, Mohanty P, editors. 2000. Probing Photosynthesis: Mechanisms, Regulation, and Adaptation. London: Taylor & Francis. p. 9-39. Retrieved Feb. 27, 2012, from http://www.life.illinois.edu/govindjee/papers/milestones.html#intro.
MATHEWS CK, VAN HOLDE KE. 1990. Biochemistry. Redwood City, CA: The Benjamin/Cummings Publishing Company, Inc. 1129 p.
MOORE R, CLARK WD, VODOPICH DS. 2003. Botany. 2nd ed. Boston, Massachusetts: McGraw-Hill. 919 p.
[US-DOE] U. S. DEPARTMENT OF ENERGY. 2012. Fossil energy: energy lessons and study guides for younger students. Retrieved Feb. 27, 2012, from http://www.fe.doe.gov/education/energylessons/index.html.WARDLAW GM, HAMPL JS, DiSILVESTRO RA. 2004. Perspectives in Nutrition. 6th ed. New York, NY: McGraw-Hill Companies, Inc. 752 p.
